Human IgM Purification Resin and Columns
IgM monoclonal antibodies have become increasingly favorable for their use as therapeutic tools, while remaining popular for their research and diagnostic applications. More IgM based therapeutics are being developed now than ever before due to a better understanding of the roles it plays in the immune system. With a molecular weight of 990,000 Daltons, IgM is the largest and hardest of the major antibodies commonly purified.
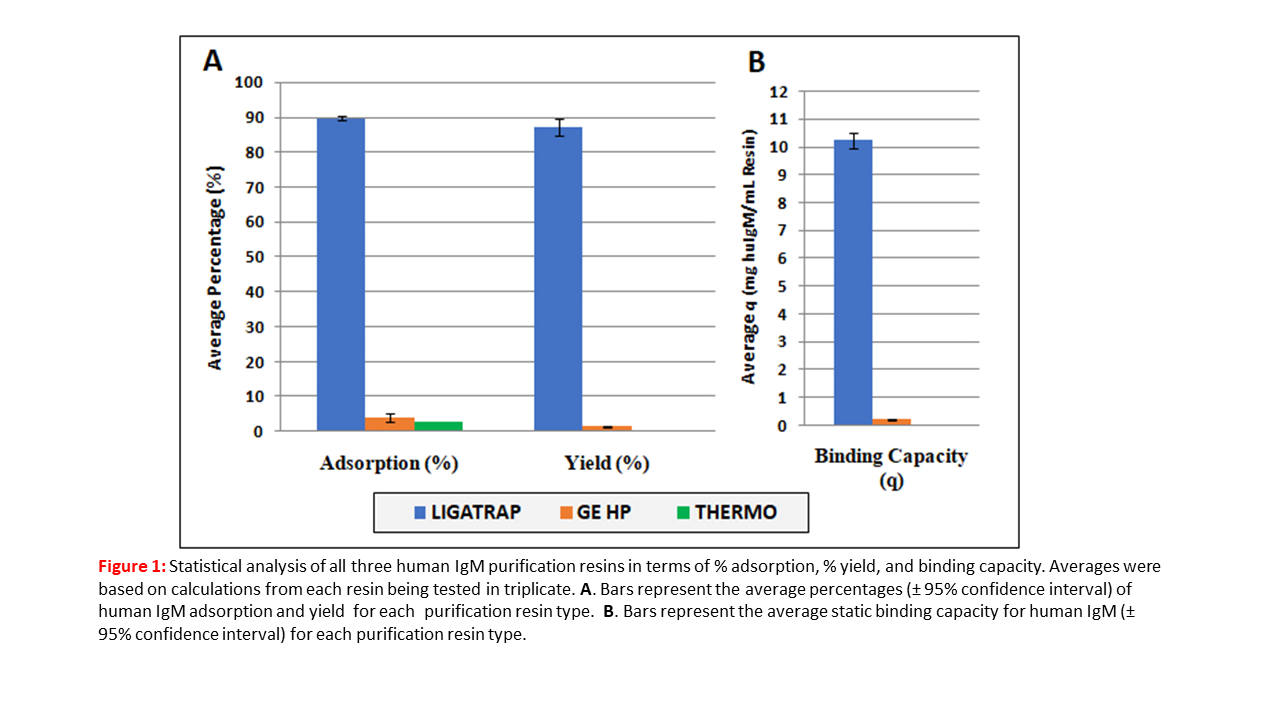
LigaTrap technology separates itself by having the highest quality chromatographic purification for IgM on the market today. The advantages LigaTrap IgM Resin include capture both heavy and light chain subtypes of IgM, no immunogenicity or leaching, mild elution conditions, and can be reused after sanitization up to 100 times. LigaTrap IgM Purification Resin is capable of processing and purifying monoclonal antibodies form cell culture supernatant, ascites fluid, hybridoma, and other sources of recombinant IgM.
Product Descriptions
Purification kits are offered in microspin column, 1mL prepacked column, and 5mL prepacked column formats. Each kit includes the appropriate buffers and protocol for performing purifications.
Prepacked columns are offered in 1mL and 5mL formats for our various lines of antibody affinity chromatography resins. Prepacked columns come in kit formats or as standalone products.
Loose resin is available in various volume sizes which can be packed for end user’s specific purification needs. For bulk resin orders larger than 100mL, please inquire.
This product is not recommended in serum purification applications due to cross-reactivity with other immunoglobulins. Please consult LigaTrap for protocols that may improve results in serum.
| Part # | Name | Amount | Price | |
| LT-143KIT | Human IgM Mircospin Column Kit w/ Buffers |
KIT-10x0.1 mL | $469.00 | Add to Cart |
| LT-143-1ml KIT | Human IgM 1mL Prepacked Column Kit w/ Buffers |
KIT-1x1 mL | $386.00 | Add to Cart |
| LT-143-5ml KIT | Human IgM 5mL Prepacked Column Kit w/ Buffers |
KIT-1x5 mL | $1,424.00 | Add to Cart |
| LT-143 | Human IgM Loose Resin-5mL |
5 mL | $842.00 | Add to Cart |
| LT-143-10ml | Human IgM Loose Resin-10mL |
10 mL | $1,056.00 | Add to Cart |
| LT-143-25ml | Human IgM Loose Resin-25mL |
25 mL | $1,515.00 | Add to Cart |
| LT-143-50ml | Human IgM Loose Resin-50mL |
50 mL | $2,522.00 | Add to Cart |
| LT-143-100ml | Human IgM Loose Resin-100mL |
100 mL | $3,783.00 | Add to Cart |
| LT-143-MSC | Human IgM Microspin Columns without buffers |
10x0.1 mL | $205.00 | Add to Cart |
| LT-143-1x1ml | Human IgM Prepacked Purification Column-1mL |
1x1 mL | $190.00 | Add to Cart |
| LT-143-1x5ml | Human IgM Prepacked Purification Column-5mL |
1x5 mL | $883.00 | Add to Cart |
| LT-143-3x1ml | Human IgM Prepacked Purification Columns-3x1mL |
3x1 mL | $545.00 | Add to Cart |
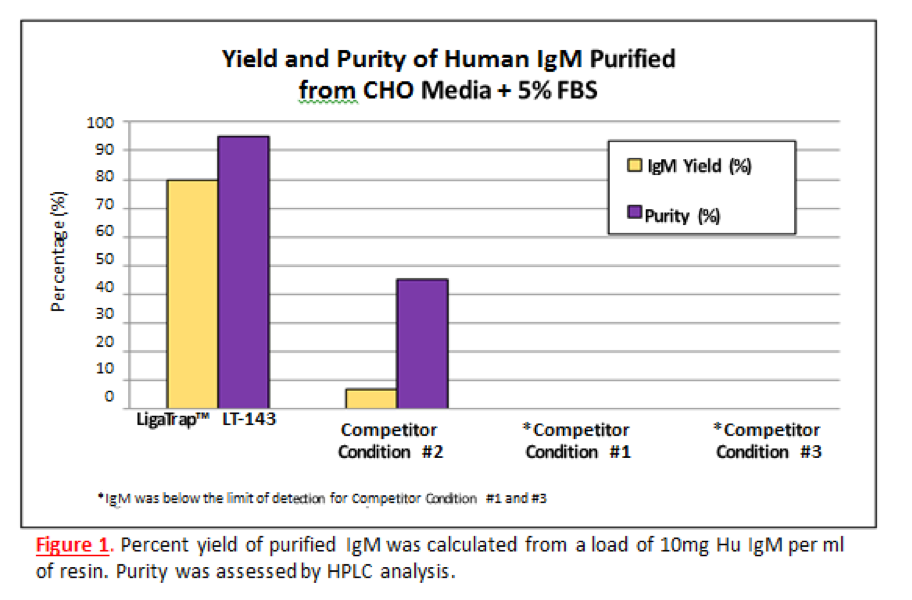
Human IgM Yield and Purity
Human IgM was purified from CHO media containing 5% FBS using and a leading competitor of IgM purification resin. Standard chromatographic conditions were used for the LigaTrap Human IgM resin. Three different manufacturer recommended binding conditions were used for the leading competitor of IgM purification resin.